
We are the employer of choice in India in the domain of Healthcare Documentation Services
Together, we improve the overall quality of care.
AQuity is India’s leading measurement-based healthcare documentation services provider for healthcare systems and practices overloaded by medical data entry and management tasks. AQuity India’s medical transcriptionists, medical coders, and virtual scribes alleviate the pressures that strict documentation requirements place on any healthcare organization. We accurately capture the complete patient narrative, allowing physicians to fully engage the patient without interruption. Take a look at some of the things that set AQuity India apart:
Our CSR Initiaive and Employee Engagement Activities
Food for Life Vehicle Handover at Coimbatore
STEM Exhibition in Airoli, Navi Mumbai
20 years of Experience
AQuity India boasts state-of-the-art infrastructure, world-class security measures, and cutting-edge processes
More than 4000 Employees
AQuity India employees make up nearly two thirds of AQuity Solutions
Global Presence
Our parent company, AQuity Solutions, extends worldwide across the U.S., the U.K., Canada, and Australia
Best-in KLAS technology and service
Among the first to adopt automated speech recognition (ASR) technology, AQuity India operates on high-end technology platforms
Secure and compliant
Our Global Delivery Centers are both HIPAA-compliant and ISO 9001:2008 certified
The right blend of technology and talent enables AQuity India to deliver daily on our promise of world-class service to our global clientele.
Industry Recognition
as the Healthcare Market Leader.

Why employees prefer AQuity?
At AQuity India, our employees are our strength. We take pride in ourselves in providing a human element to clinical data management, a critical part of any health care business. Empathy is the cornerstone of delivering the best results to our clients, and driving better outcomes for their patients. As such, our workforce is the powerhouse that makes what we do possible, and we constantly ensure that our employees are empowered.
Our employees work in many locations, but function together as a strong team made up of individuals. To ensure each employee is rewarded for their hard work, we have built a culture that provides and enforces an open door policy and encourages an open dialogue across all departments. In the best interests of the company and its employees, we ensure that only the best talent is hired and the infrastructure, facilities, exposure is such, that it allows continuous learning and progress both as an individual and as a professional.
These values extend across our India offices and across the globe, ensuring that all employees share opportunities for growth, alignment on expectations, and a close team structure.

Interested in joining AQuity India? Locate or contact an office near you.

Our teams are led by an array of seasoned professionals. Discover the people behind AQuity.
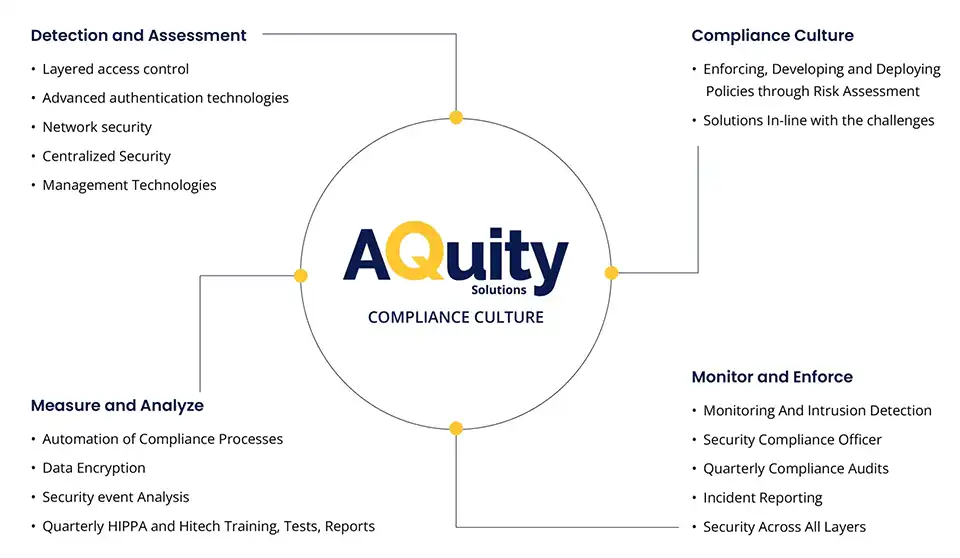
Security and Sophistication
AQuity India’s approach to handling sensitive healthcare data relies heavily on a multi-pronged, layered approach to safety and compliance. It is the crux of establishing trust with our clients and maintaining compliance with regulations across the world.
Take a look at our process approach:
Interested in working at AQuity?
for careers information or browse our current openings.







